If CO2 is LOW, it means there are not enough respiratory acids because he's probably hyperventilating too much CO2 away. If your pH is acidic, and your CO2 is HIGH, its considered respiratory acidosis. If CO2 is HIGH, it means there is a buildup of respiratory acids because he's not breathing enough CO2 away. If you measured venous blood gasses, the levels are different and PvCO2 is used. How much respiratory acid (CO2) should there be? The normal value is 35-45 mmHg (mmHg just means millimeters of mercury, its a measurement of pressure.) The (a) in PaCO2 just stands for arterial. We use arterial blood gas tests to check it. We can measure it to see how much respiratory acid (CO2) there is in the blood. PaCO2 is the partial pressure of carbon dioxide. We measure the pH, the amount of carbon dioxide ( PaCO2) and the amount of oxygen in the blood ( PaO2). They measure the amount of each gas in your blood. We can measure the amount of respiratory acid in the arterial blood using blood gases. The extra CO2 molecules combine with water in your body to form carbonic acid and makes your pH go up. 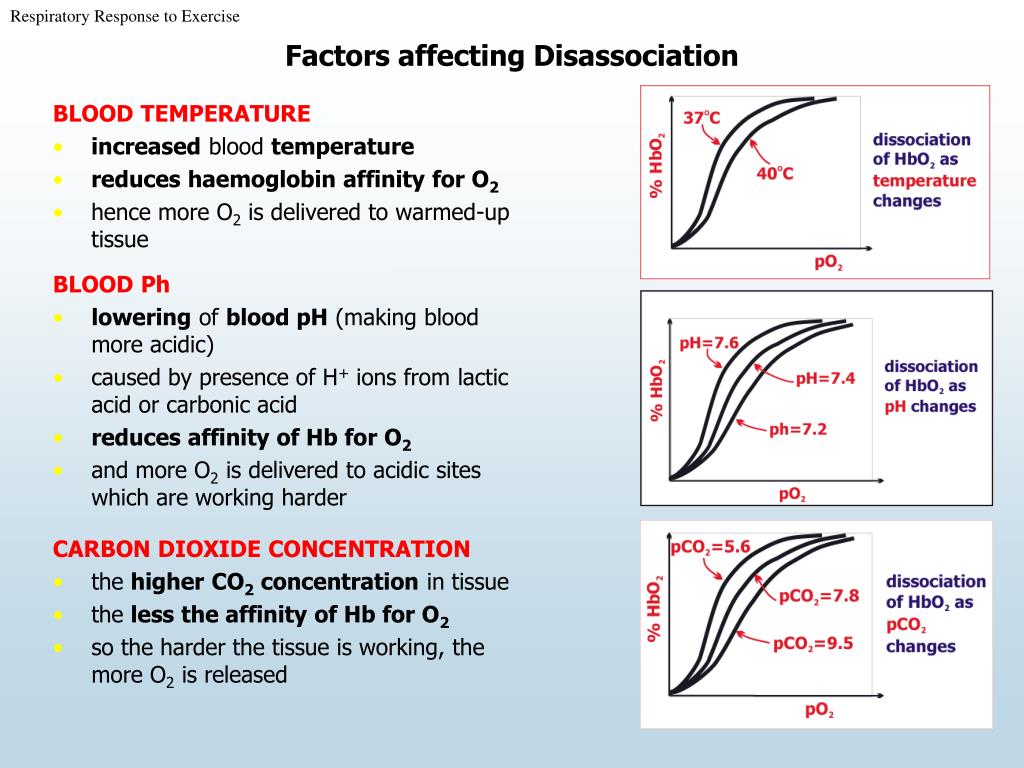
You take in oxygen by inhaling, your body turns oxygen into carbon dioxide, you exhale and remove the carbon dioxide from your body.Ĭarbon dioxide is "respiratory acid." When you're not breathing adequately, you are not getting rid of this "respiratory acid" and it builds up in the tissues. pH higher than 7.45 is considered Alkaline. NORMAL - Just right! pH between 7.35 and 7.45 is perfect for homeostasis.ĪLKALINE- NOT ENOUGH ACID - It just means NOT acidic enough. 
This is all Acid-Base balance means.ĪCIDIC - TOO MUCH ACID - Lower than 7.35 means that there is too much acid in your system. You want to keep the body neither too acidic or too alkaline, you want to balance it. So, when we find someone whose acid levels are not conducive with life, we counteract the problem and restore homeostasis. 0.5 in either direction away is not conducive to life. The pH scale goes from 0-14, so neutral is 7 (water is neutral.) The body needs a pH of 7.35-7.45 to maintain homeostasis. When referencing acidity, we're talking about excess hydrogen ions (H+), but we're just going to use the general term "acid." Its a scary foreign phrase, but it just means "percentage of acids" Acid isn't always a bad thing like in movies. It just measures how acidic something is. This causes the equilibrium to shift to the right, the H 3O + concentration increases and pH drops.Ĭontinue and read about the metabolic problems that can result in blood pH changes.PH is a measurement of acidity. 
A hypoventilating (excessively shallow breathing) person does not expel enough carbon dioxide and has elevated blood carbon dioxide levels. Respiratory acidosis is caused by the reverse process. In doing so, they rebreathe some of expelled carbon dioxide, and blood carbon dioxide levels return to normal. Respiratory alkalosis is treated by having the hyperventilating person breathe into a paper bag. The corresponding drop in H 3O + concentration causes an increase in pH.Ģ H 2O + CO 2 H 2CO 3 + H 2O H 3O + + HCO 3. As a result the carbon dioxide concentration in the blood is reduced and the bicarbonate/carbonic acid equilibrium shifts to the left. When a person hyperventilates they exhale more carbon dioxide than normal. The increase in pH is often caused by hyperventilation (excessively deep breathing). Respiratory alkalosis is a condition in which the pH of the blood is above normal. Blood pH can be disturbed by a number of respiratory and metabolic causes. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
